The KLAS Digital Pathology 2026 Report reveals a significant shift in the U.S. digital pathology market, driven by recent FDA clearances and advancements in reimbursement strategies. Despite this growth, the adoption of digital pathology solutions remains in its infancy, with fewer than 15% of healthcare organizations in the United States having selected a digital pathology vendor.
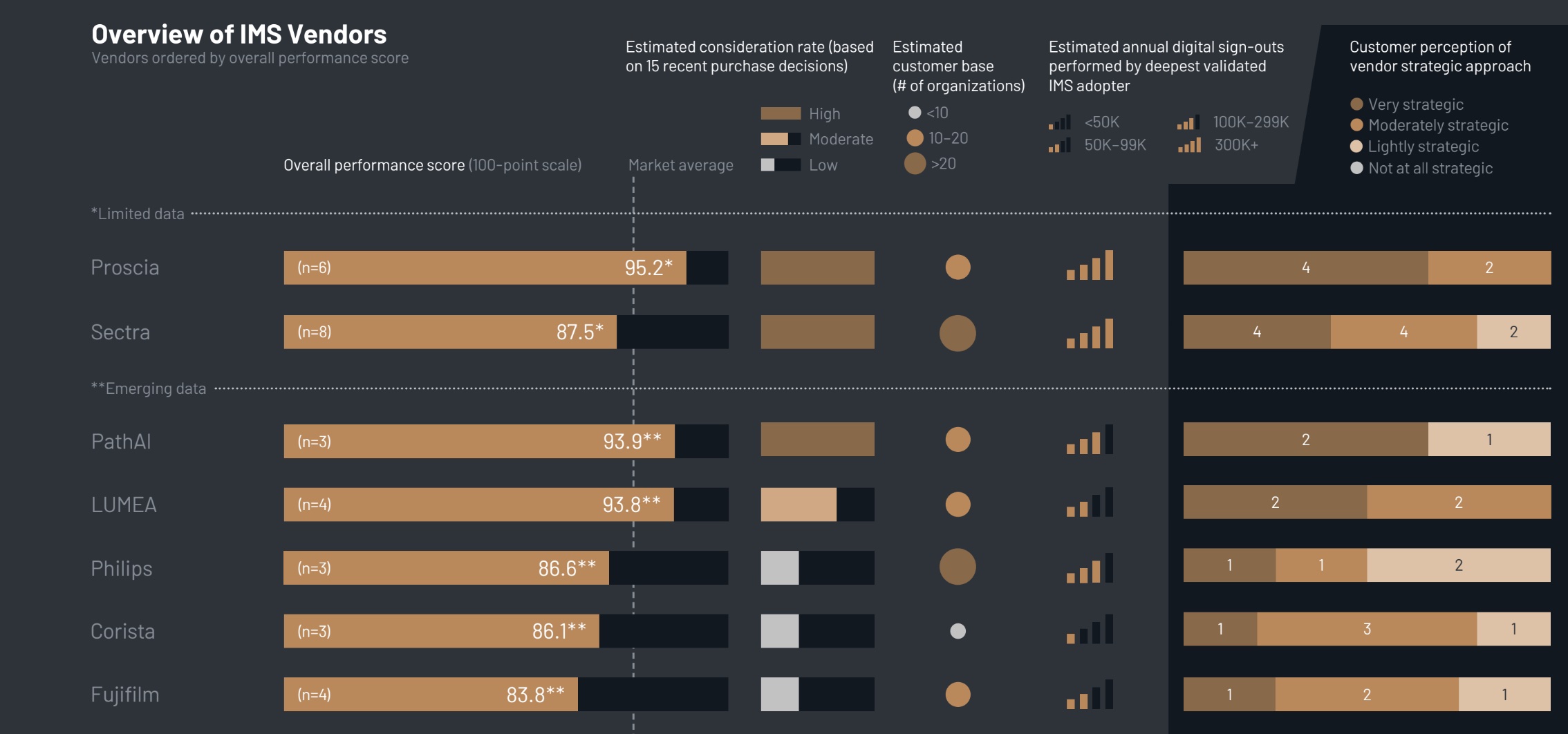
Emerging Leaders in Image Management Systems
When evaluating Image Management Systems (IMS), vendors specializing in pathology, such as Proscia, PathAI, and LUMEA, are outpacing traditional radiology providers in customer satisfaction. Users commend these vendors for their tailored platforms and strategic support, which are designed specifically for the needs of pathology.
In contrast, traditional imaging vendors like Sectra, Philips, and Fujifilm are expanding into the pathology space but are facing challenges. Customers often report a desire for more proactive guidance, especially regarding the integration of artificial intelligence (AI) within their workflows.
Proscia stands out as the most frequently considered IMS vendor, with its collaborative, education-focused approach being a notable differentiator. Initial feedback on PathAI highlights its proactive communication and hands-on integration efforts, alongside strong engagement from its leadership. Customers of LUMEA praise the system’s user-friendliness and the vendor’s commitment to adapting workflows to meet specific needs.
Dominance in Scanner Hardware
In the realm of scanner hardware, Leica Biosystems is recognized as the dominant force in the U.S. market. Its Aperio GT 450 DX scanner, one of the first to receive FDA clearance, is widely adopted for clinical use. Customers consistently emphasize its high-quality imaging, efficient slide loading, and reliability as key factors that drive productivity.
Conversely, users of Roche scanners express significant dissatisfaction, citing reliability issues and low throughput that hinder high-volume diagnostic workflows. This feedback aligns with global research trends, indicating a pressing need for improvement in Roche’s scanner capabilities.
The integration of AI into digital pathology processes is also gaining traction. Hospitals are increasingly evaluating AI tools for clinical applications, particularly algorithms related to breast and prostate cancer. A critical requirement for these technologies is seamless integration into existing IMS workflows, enabling pathologists to utilize AI without navigating separate systems.
The focus on breast cancer applications, particularly those aiding in biomarker assessment such as Ki-67, HER2, estrogen receptor (ER), and progesterone receptor (PR), highlights the urgency and relevance of AI in this field. Key developers in this space include Ibex, Visiopharm, Paige.ai, and PathAI, all of which are under consideration by healthcare organizations looking to enhance their diagnostic capabilities.
As the digital pathology market evolves, the distinction between specialist and generalist vendors remains clear. Specialist vendors, equipped with a focused approach, are currently leading in customer satisfaction. This trend suggests a future where tailored solutions in digital pathology may reshape how healthcare organizations approach diagnostics and patient care.
For a deeper dive into the findings of the KLAS Digital Pathology 2026 Report, visit KLAS Research.